|
Plots of pressure vs temperature for three different gas samples extrapolate to absolute zero. The constant volume gas thermometer plays a crucial role in understanding how could be discovered long before the advent of. Consider a graph of pressure versus temperature made not far from standard conditions (well above absolute zero) for three different samples of any ideal gas (a, b, c). To the extent that the gas is ideal, the pressure depends linearly on temperature, and the extrapolation to zero pressure occurs at absolute zero.
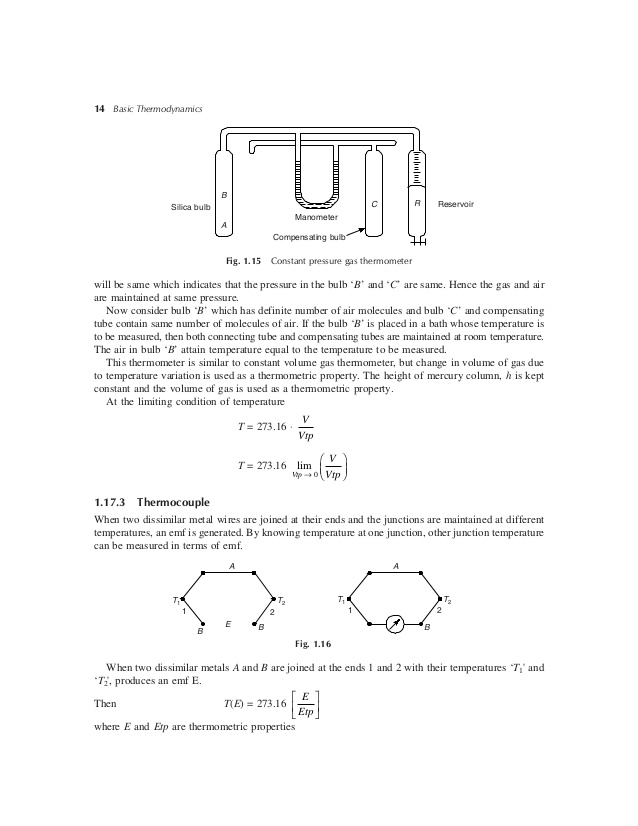
Temperature Ranges of Application 3. Constant Volume Thermometer 4. Thermocouples 5. Thermometer Time. Temperature •Pressure is a force applied to an area, so its measure. Separated by the H2 gas from a constant T heat-sink Metal film is both heater and thermometer Qtotal=QH2 conduction. Thermometer may be chosen to be the change in pressure of a gas kept at constant volume, or the change in volume of a gas kept at constant pressure. Since it is easier to measure accurately change in pressure than it is to measure change in volume, constant-volume gas thermometers are more common in use than constant-pressure gas thermometers.
Note that data could have been collected with three different amounts of the same gas, which would have rendered this experiment easy to do in the eighteenth century.
Comments are closed.
|
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
- Home
- About Brooke
- Blog
- Ead Theatermaster 8000 Pro Manual
- Download Aqua Real 3D Deluxe 4.01 Crack
- Midland 77-285 Manual
- Download Whatsapp For Android 2.3.6
- 2007 Chevrolet Silverado Southern Comfort Edition
- Lg Flatron M2262d Software Update
- Kevin Finel Auto Hypnose Pdf Merge
- The Rifles No Love Lost Album Download
- Contact
- Xenosaga Ds Rom English Patch
- Sims 4 melanin pack 3
- Igo primo maps 2018
- Vlc media player download window 7
- Wii remote apk download
- Fxp to wav converter
- Curseforge download minecraft
- Gta 3 apk mediafire
- GTA V Zip Andorid Ap
- Monster musume hentai
- The sims 3 cc prison
- Adobe flash cs6 serial number list
- Sims 3 cc folders download
- Discord ip grabber plugin
- Warcraft 2 map editor not letting me play custom games
- Critical ops online fps games android
- Coolorus free download full
- I doser doses
- Wic reset utility download full version
- Bloody roar 2 download for pc
- Magic snow plugin c4d download free
- The sims 3 cc clothes skirts
- Can you download zoom on a chromebook
- Yaesu Ft 757Gx Problems
- How to get adobe flash cs6 free
- My free mp3 song download juice
- Harvest moon tree of tranquility rom emuparadise
- Download buku erlangga gratis sd
 RSS Feed
RSS Feed
